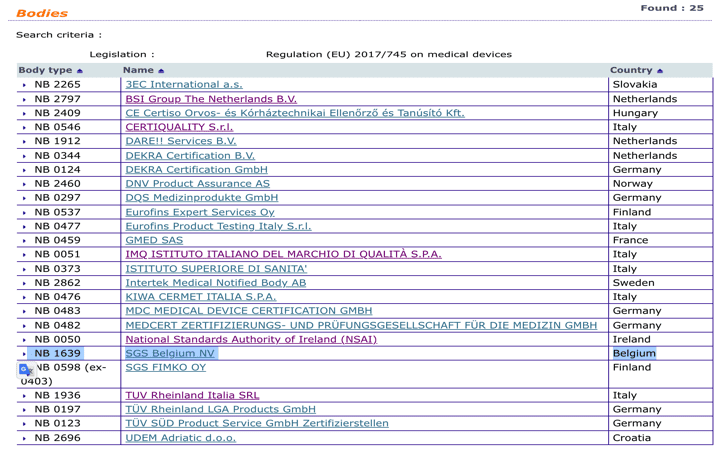
Informational - New designated Notified Body for Regulation (EU) 2017/745 ( MDR) included in Nando – IMQ ISTITUTO ITALIANO DEL MARCHIO DI QUALITÀ S.P.A.

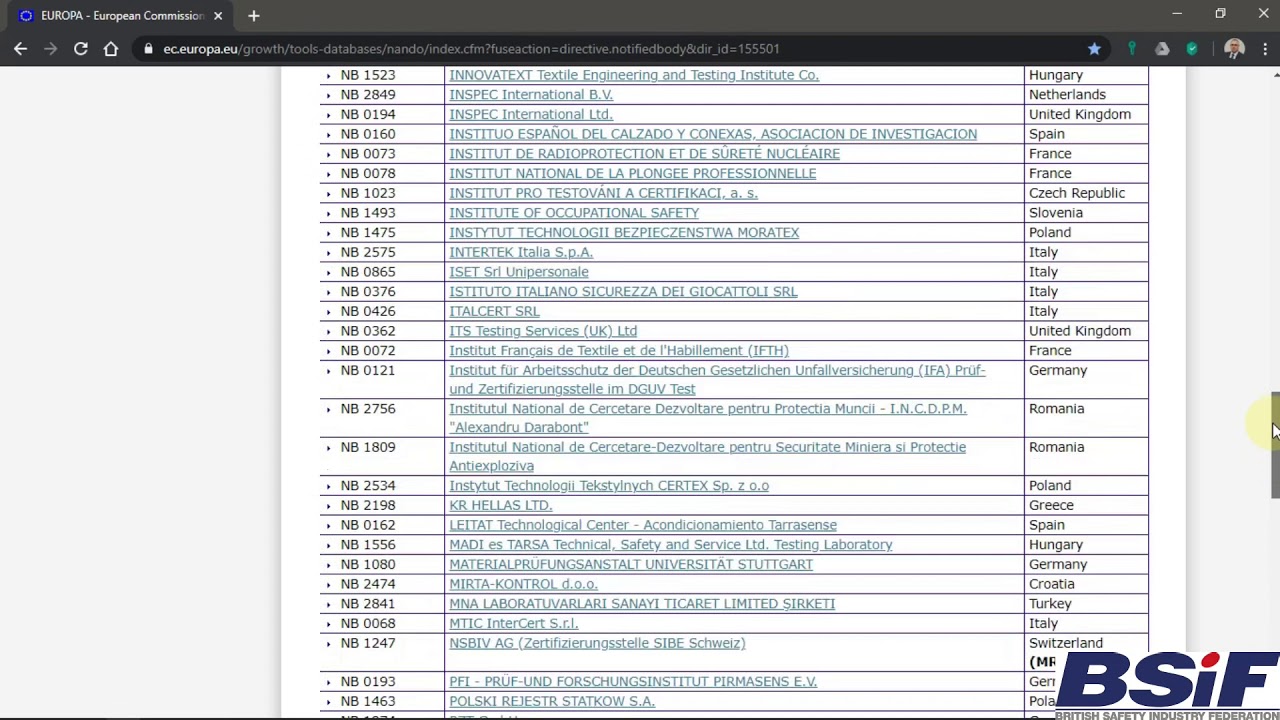
MDR: 29 Notified Bodies on NANDO · MDlaw – Information platform on European medical device regulations
![Glossary] EU MDR and IVDR: The core terms you should know - Pharma Serialization, Aggregation and Track and Trace Software by SoftGroup Glossary] EU MDR and IVDR: The core terms you should know - Pharma Serialization, Aggregation and Track and Trace Software by SoftGroup](https://www.softgroup.eu/wp-content/uploads/2020/10/Untitled-design-38-1280x720.png)
Glossary] EU MDR and IVDR: The core terms you should know - Pharma Serialization, Aggregation and Track and Trace Software by SoftGroup

MDR: 26 Notified Bodies on NANDO & Swiss economic operator's requirements updated! · MDlaw – Information platform on European medical device regulations

Amazon | CHPO シーエイチピーオー 腕時計 チーポ NANDO SLAYER MESH 40mm ブラック 14232QQ メンズ レディース 正規輸入品 | 腕時計 | ファッション 通販
Dear Client, Regulation (EU) 2017/745 of the European Parliament and of the Council (MDR) - replacing Directives 93/42 / EEC (MD

Organismos Notificados IVDR (8): QMD Services (Austria) ON num. 2962 nuevo ON. Enhorabuena !!! | Red de Tecnologías Sanitarias y Productos Sanitarios
![Xplood]081 | by ɳɑɳɖɵ ɑɾɾʉɗɑ Nando Arruda © All rights rese… | Flickr Xplood]081 | by ɳɑɳɖɵ ɑɾɾʉɗɑ Nando Arruda © All rights rese… | Flickr](https://live.staticflickr.com/4023/4701505689_851b495793_b.jpg)

![EU MDR Quality Management System [Role of an eQMS Software] EU MDR Quality Management System [Role of an eQMS Software]](https://www.simplerqms.com/wp-content/uploads/2022/11/eu-mdr-notified-bodies-database.jpg)









![BSI News] BSI Netherlands Notified Body (NB 2797)도 MDR 심사 기관으로 지정되었습니다! | BSI Blog BSI News] BSI Netherlands Notified Body (NB 2797)도 MDR 심사 기관으로 지정되었습니다! | BSI Blog](http://bsiblog.co.kr/wp-content/uploads/BSI-Netherland-MDR.png)