physical chemistry - How do you calculate the heat of a reaction given a table of heat of formation values? - Chemistry Stack Exchange

q calculate the volume occupied at 270 c and at 2 atm pressure of the gas evolved by 2ml of solid co2 given density of solid co2 is 153 g ml 1

OneClass: You want to extract a solute from 100 mL of water using only 300 mL of ether. The partition...

A solution contains 5.6 mL of alcohol mixed with 75 mL of water. Calculate the concentration - YouTube
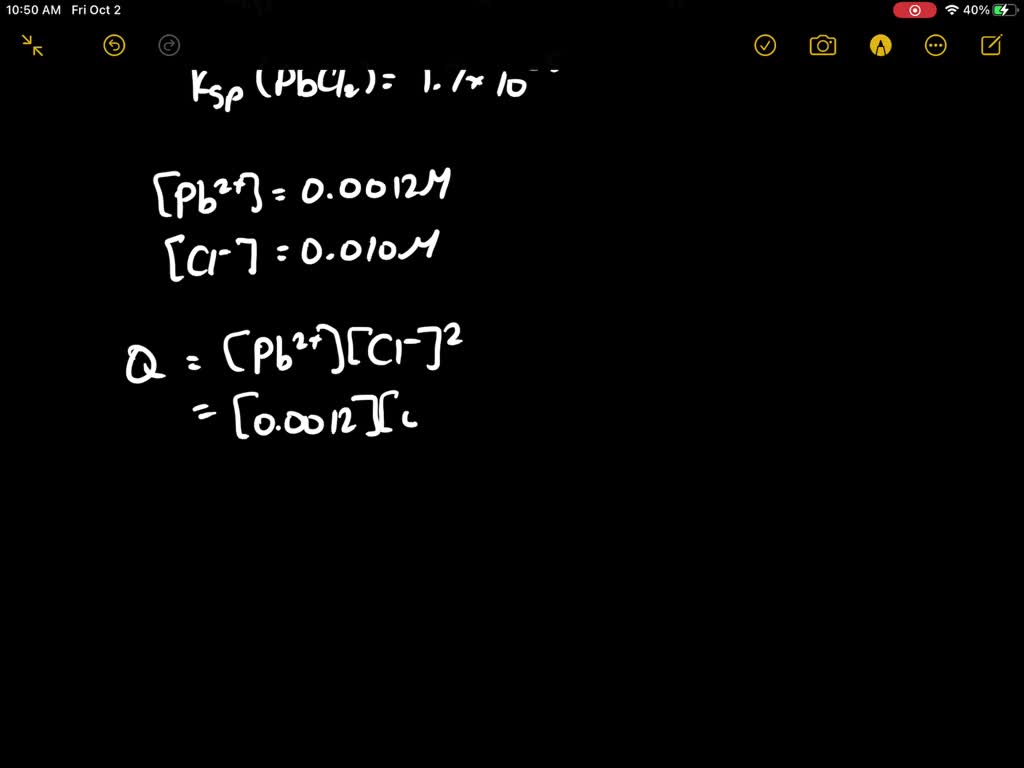
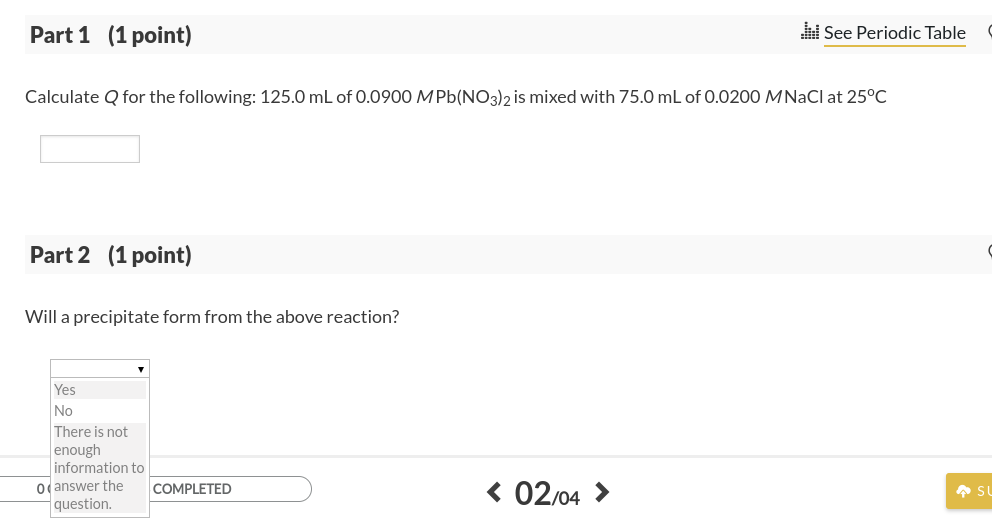
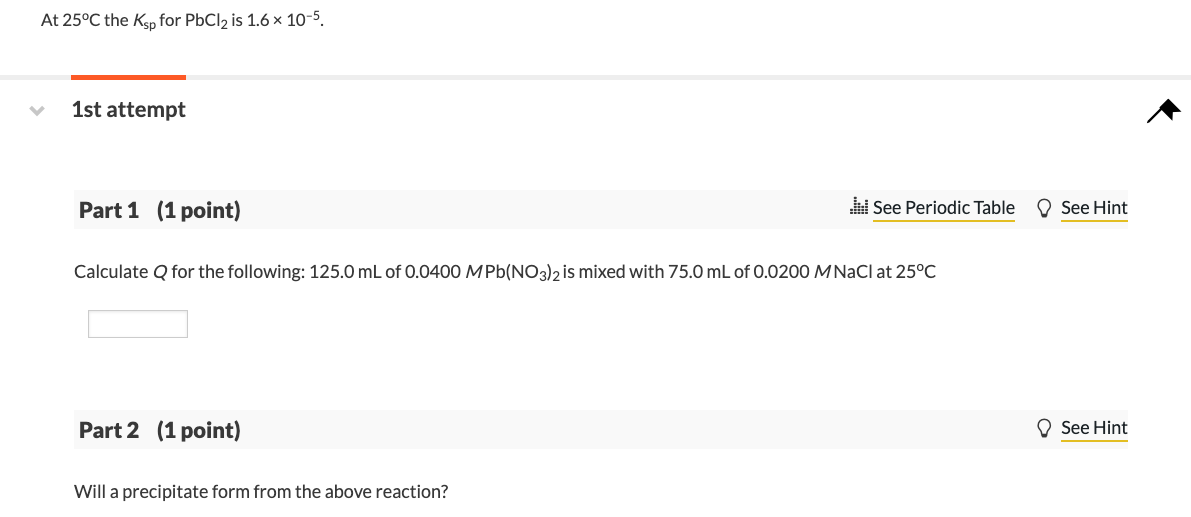
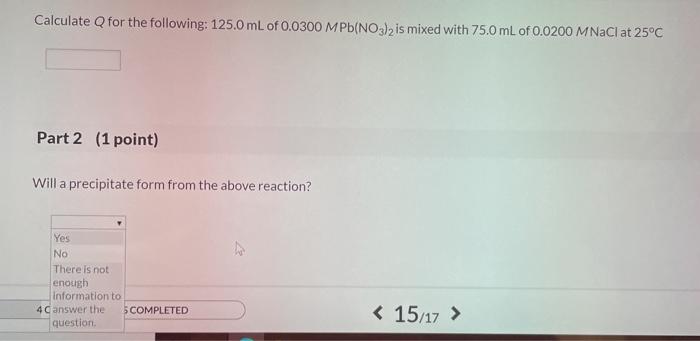
SOLVED: At 25oC the Ksp for PbCl2 is 1.6 × 10–5. A. Calculate Q for the following: 125.0 mL of 0.0800 M Pb(NO3)2 is mixed with 75.0 mL of 0.0200 M NaCl