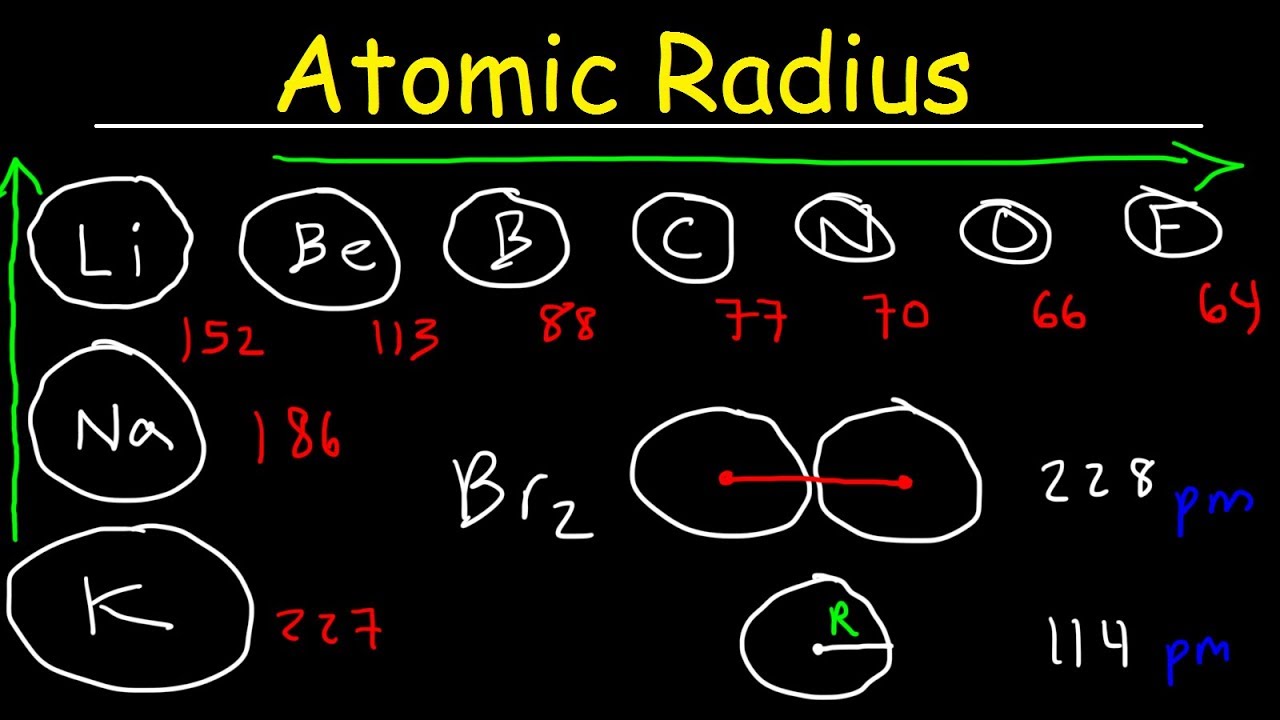
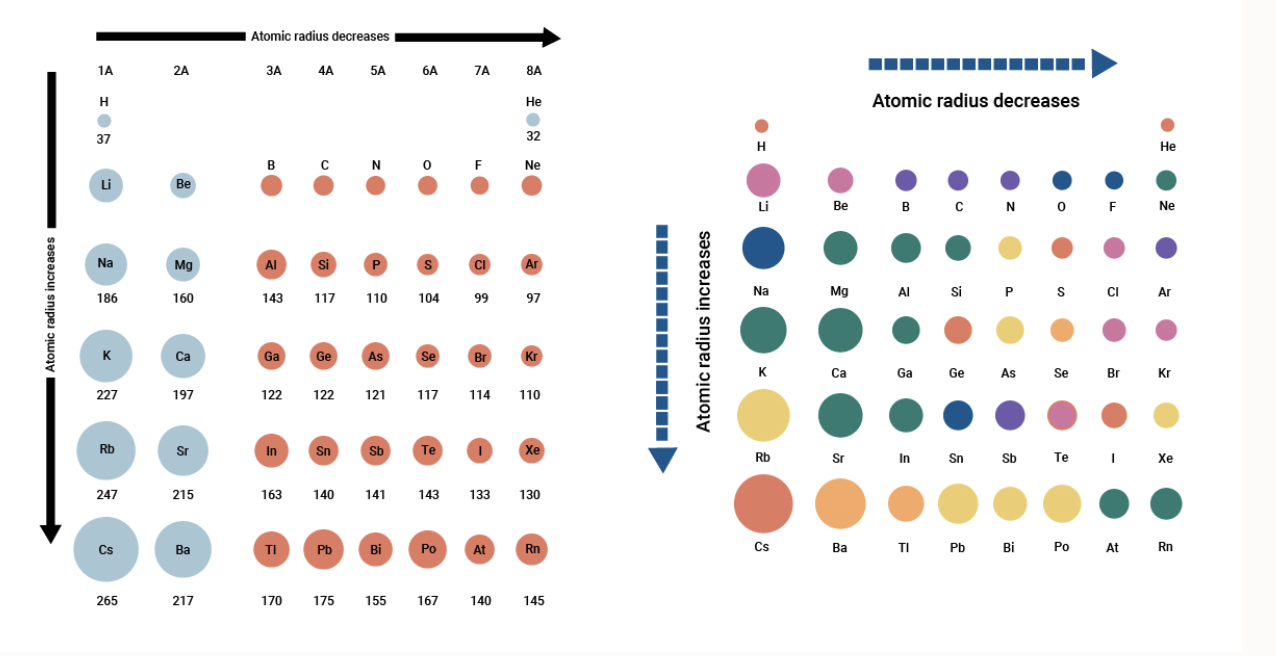
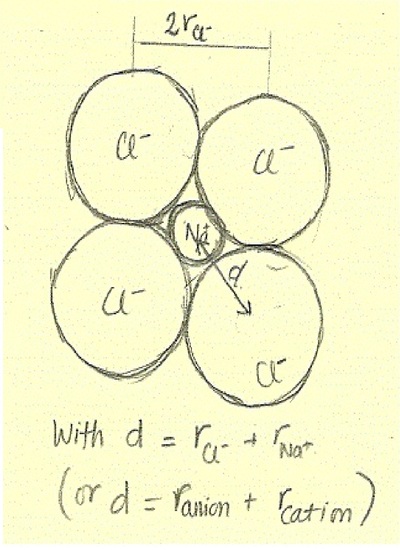Atomic radius and ionic radius of F(g) and F(g)^- are 72 and 136 pm prespectivley. Calculate the ratio and percentage increase in terms of volume during formation of F(g)^(-) form F(g) .
Derive a formula for radius of the stable orbit of hydrogen atom on the basis of Bohr model. Prove that in hydrogen - Sarthaks eConnect | Largest Online Education Community

Niobium has a density of 8.57 g/cm3 and crystallizes with the body-centered cubic unit cell. Calculate the radius of a niobium atom - Chemistry Stack Exchange

Atomic radius of `Li is 1.23 Å` and ionic radius of `Li^(+)` is `0.76 Å`. Calculate the percentage - YouTube
Derive a formula for radius of the stable orbit of hydrogen atom on the basis of Bohr model. Prove that in hydrogen - Sarthaks eConnect | Largest Online Education Community

Atomic radius is of order 10^-8 cm and nuclear radius is of order 10^-13. calculate what fraction of atom is occupied by nucleus? | Socratic

Calculate the radius of second Bohr orbit in hydrogen atom from the given data.Mass of electron = 9.1 × 10^-31Kg Charge on the electron = 1.6 × 10^-19C Planck's constant = 6.63 ×